Tuesday, February 28, 2006
Medical Waste
Many of you probably saw the "60 Minutes" segment on human embryonic stem cell research. One especially maddening aspect of the anti-stem cell argument is that thousands of excess embryos are discarded at fertility clinics, yet they cannot be used for life-saving research in federally-funded labs. This cartoon from the Buffalo News really nails it


Monday, February 27, 2006
Sunday, February 26, 2006
Findings of Scientific Misconduct, Part I

Medical research ethics is a soapbox of mine. As part of what I plan as a continuing series, I will periodically post particularly egregious cases of scientific misconduct. These are public notices sent to those of us on the mailing list of the National Institutes of Health. (I have condensed the announcement for the sake of brevity). As you read this, keep in mind that people's lives and taxpayer dollars were entrusted to these study coordinators. These kind of misdeeds give a black eye to the medical research field and make my blood boil.
Notice Number: NOT-OD-06-042
Key Dates Release Date: February 24, 2006
Issued by Department of Health and Human Services (DHHS), (http://www.hhs.gov/)
Notice is hereby given that the Deputy Assistant Secretary for the Office of Acquisition Management and Policy of the Department of Health and Human Services (HHS) has taken final agency action in the following case:
Paul H. Kornak, Stratton VA Medical Center, Albany, New York
Of the 48 criminal charges contained in his Indictment, Paul Kornak pled guilty to three criminal charges. See United States of America v. Paul H. Kornak, Criminal Action No. 03-CR-436 (FJS), U.S. District Court (N.D.N.Y.) (January 18, 2005). In addition to the 71-month term of imprisonment imposed, Mr. Kornak was directed to pay restitution to two pharmaceutical companies and the VA in the amount of approximately $639,000. As part of his guilty plea, Mr. Kornak admitted to the following facts:
In August 2000, Mr. Kornak applied for employment to the VA, submitting a false ``Declaration for Federal Employment'' form. Mr. Kornak denied that he had been convicted or on probation in the preceding 10 years, whereas in fact, he had been convicted of mail fraud in 1992 and placed on probation for 3 years.
By October of 2000, Mr. Kornak was responsible for organizing, coordinating, implementing, and directing all research elements in the Stratton VA Medical Center oncology research program. Specifically, Mr. Kornak was the site coordinator at the Stratton VA Medical Center for the ``Iron (Fe) and Atherosclerosis Study'' (FeAST), cancer studies known as Tax 325 and Tax 327, and a bladder cancer study. The FeAST study was a clinical trial that tested a novel procedure for controlling atherosclerosis, also known as hardening of the arteries, by reducing the iron in the body through blood drawing. The Tax 325 cancer treatment study involved the administration of pharmaceutical products to patients with metastatic or locally recurrent gastric cancer previously untreated with chemotherapy for advanced disease. The Tax 327 study involved the administration of pharmaceutical products to patients with metastatic hormone refractory prostate cancer. The purpose of the bladder cancer study, which was co-sponsored by the National Cancer Institute, National Institutes of Health, was to compare the use of difluoromethylornithine (DFMO) to the use of a placebo in patients with low grade superficial bladder cancer according to time to first recurrence of the tumor and toxicities.
From May 14, 1999, to July 10, 2002, in connection with the above protocols, Mr. Kornak participated in a scheme to defraud the sponsors of the clinical studies in that ``he would and repeatedly did submit false documentation regarding patients and study subjects and enroll and cause to be enrolled persons as study subjects who did not qualify under the particular study protocol.''
Mr. Kornak caused the death of a study subject when he ``failed to perceive a substantial and unjustifiable risk that death would occur when he knowingly and willfully made and used . . . documents falsely stating and representing the results of [the study subject's] blood chemistry analysis . . ., which false documents purported that [the study subject] met the inclusion and exclusion criteria for participation in Tax 325 when the actual results did not meet the inclusion and exclusion criteria and showed impaired kidney and liver function, and [the study subject] thus was administered the chemotherapeutic drugs docetaxel, cisplatin, and 5-FU in connection with Tax 325 on or about May 31, 2001, and died as a result thereof on or about June 11, 2001.'' Based on the criminal conviction and the facts admitted to above, HHS and VA believe that a debarment period longer than the standard length of debarment is warranted in this case.
Mr. Kornak admitted to a dishonest handling of the research records and demonstrated a complete disregard for the well-being of vulnerable human subjects under his care. In pleading guilty to criminally negligent homicide, Mr. Kornak admitted that a reasonable person would have perceived a substantial and unjustifiable risk of death if an ineligible subject were enrolled in the cancer study in question and that his failure to perceive such a risk in enrolling the ineligible subject constituted a gross deviation from the standard of care. Moreover, a longer debarment period is warranted in this case because of an established pattern of misconduct and criminal behavior on the part of Mr. Kornak. As stated above, Mr. Kornak has a prior conviction of mail fraud. In addition, the Office of Personnel Management excluded Mr. Kornak from all Federal nonprocurement transactions for an indefinite period, effective July 22, 1993. Nonetheless, beginning in 1999, Mr. Kornak actively participated in federally sponsored research protocols in violation of the imposed exclusion.
A lifetime debarment of Mr. Kornak is necessary to protect the public interest overall. Given the scope of his criminal conviction, his longstanding pattern of criminal behavior, and his total disregard for the safety and well-being of human subjects, Mr. Kornak's responsibility to engage in transactions with the Federal Government cannot be assured at any time in the future.
Stem Cell Central
I get a lot of questions about the sort of projects that are going on at Stem Cell Central.
Here is a bit about what we do:
Stem Cells in the Eye
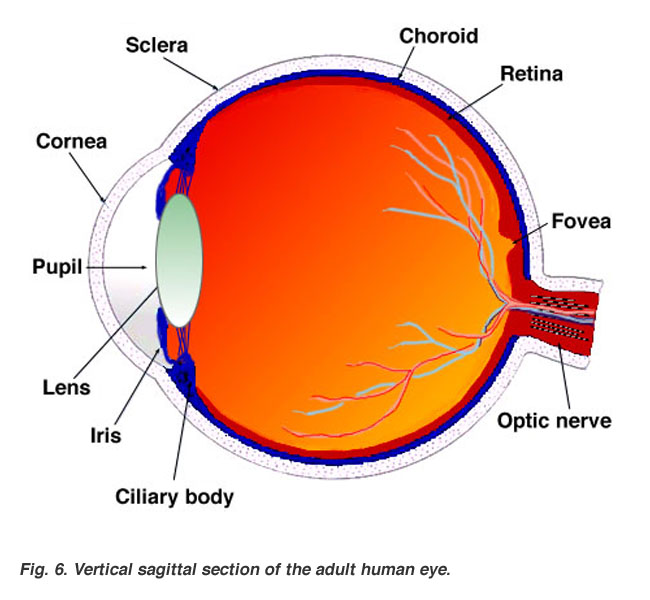
Stem cells can be found in both embryos and adults. They are the original building blocks for every organ, tissue and cell in the body, including the eye. Stem cells behave like blank microchips that can be programmed to perform any number of specialized tasks. My laboratory studies stem cells in the eye, as a way to repair and to prevent damage to the eye. We have two important stem cell projects going on at Stem Cell Central. These involve the cornea and the retina, depicted in the diagram below:

Here is a bit about what we do:
Stem Cells in the Eye
Stem cells can be found in both embryos and adults. They are the original building blocks for every organ, tissue and cell in the body, including the eye. Stem cells behave like blank microchips that can be programmed to perform any number of specialized tasks. My laboratory studies stem cells in the eye, as a way to repair and to prevent damage to the eye. We have two important stem cell projects going on at Stem Cell Central. These involve the cornea and the retina, depicted in the diagram below:

The Cornea:
The cornea is the clear outermost surface of the eye and is subject to trauma, injury, and every day wear and tear. The stem cells of the cornea are located at the limbus (the thin area between the clear cornea and the white sclera of the eye). The purpose of corneal, or limbal, stem cells is to maintain a healthy eye surface and replace cells that are lost and blinked away. Limbal stem cells can be transplanted for severe eye injuries. During this outpatient procedure, some limbal stem cells are taken from the healthy eye, or a donor's eye, and then transplanted to the affected eye.
In a successful transplant, limbal stem cells will produce a new healthy layer of cells in the patient's eye. The success rate varies from 25 percent to 70 percent, depending on the underlying condition of the affected eye. Limbal stem cell transplantation in the eye holds great promise for patients. We are still working to determine the best source of cells to use, the best way to transfer the cells and better ways to decrease the risk of rejection between donor cells and the recipient.
Retinoblastoma: A cancer of the eye
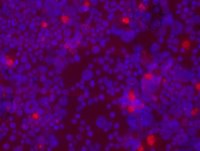
Retinoblastoma is the most common eye tumor of early childhood, with 90% of cases occurring before the age of five. This early onset of disease led us to investigate the earliest origins of retinoblastoma, in the form of primitive stem cells. My laboratory, has identified cancer stem cells in retinoblastoma tumors that are believed to be responsible for the spread of tumors as well as resistance to chemotherapy. Although cancer stem cells comprise a small percentage of the total tumor, it only takes one pesky stem cell to spread the tumor to another location in the body. The good news is that we can now target these cancer stem cells for new treatments that will circumvent their unique resistance to chemotherapy and their ability to spread tumors to distant sites. You can see these stem cells as the red dots in a sea of blue in this recent photomicrograph from my laboratory:
The cornea is the clear outermost surface of the eye and is subject to trauma, injury, and every day wear and tear. The stem cells of the cornea are located at the limbus (the thin area between the clear cornea and the white sclera of the eye). The purpose of corneal, or limbal, stem cells is to maintain a healthy eye surface and replace cells that are lost and blinked away. Limbal stem cells can be transplanted for severe eye injuries. During this outpatient procedure, some limbal stem cells are taken from the healthy eye, or a donor's eye, and then transplanted to the affected eye.
In a successful transplant, limbal stem cells will produce a new healthy layer of cells in the patient's eye. The success rate varies from 25 percent to 70 percent, depending on the underlying condition of the affected eye. Limbal stem cell transplantation in the eye holds great promise for patients. We are still working to determine the best source of cells to use, the best way to transfer the cells and better ways to decrease the risk of rejection between donor cells and the recipient.
Retinoblastoma: A cancer of the eye
Retinoblastoma is the most common eye tumor of early childhood, with 90% of cases occurring before the age of five. This early onset of disease led us to investigate the earliest origins of retinoblastoma, in the form of primitive stem cells. My laboratory, has identified cancer stem cells in retinoblastoma tumors that are believed to be responsible for the spread of tumors as well as resistance to chemotherapy. Although cancer stem cells comprise a small percentage of the total tumor, it only takes one pesky stem cell to spread the tumor to another location in the body. The good news is that we can now target these cancer stem cells for new treatments that will circumvent their unique resistance to chemotherapy and their ability to spread tumors to distant sites. You can see these stem cells as the red dots in a sea of blue in this recent photomicrograph from my laboratory:
In summary.....
Stem cells are the way of the future. They have exciting potential to repair damage to the eye and are already being used for repair of the cornea. Adult stem cells, such as the ones we use from the corneas of consenting organ donors, are not under federal restrictions and cause no ethical concerns. Everyone can be enthusiastic about the idea of using these cells to repair and replace damaged cells of the eye in a variety of dieases. The possibilities are endless.
Stem cells are the way of the future. They have exciting potential to repair damage to the eye and are already being used for repair of the cornea. Adult stem cells, such as the ones we use from the corneas of consenting organ donors, are not under federal restrictions and cause no ethical concerns. Everyone can be enthusiastic about the idea of using these cells to repair and replace damaged cells of the eye in a variety of dieases. The possibilities are endless.
Saturday, February 25, 2006
Welcome to Raging Coconuts!
Greetings!
It will come as no surprise to many of you that this blog is going to be dedicated to the world of science and scientific research. However, my "Raging Coconuts" theory states that a thread title often has no relationship to the comments contained therein. Therefore, I fully expect to see plenty of humor and punnery along with the serious comments.
So, to get us started, I am posting this thread to welcome you all. Please feel free to leave comments about the blog format, as well. I am new to this venue and will probably make some beginners' mistakes.
Sit back, kick up your feet and sit a spell. I welcome you all as friends.
It will come as no surprise to many of you that this blog is going to be dedicated to the world of science and scientific research. However, my "Raging Coconuts" theory states that a thread title often has no relationship to the comments contained therein. Therefore, I fully expect to see plenty of humor and punnery along with the serious comments.
So, to get us started, I am posting this thread to welcome you all. Please feel free to leave comments about the blog format, as well. I am new to this venue and will probably make some beginners' mistakes.
Sit back, kick up your feet and sit a spell. I welcome you all as friends.
Subscribe to:
Posts (Atom)